SWIFT Volume Imaging Resources
Observing and controlling volumetric dynamics at cellular and near-cellular resolution
While it is clear that coordinated activity between many neurons spanning large volumes of intact tissue is critical to complex animal behavior, our understanding of such systems and their dynamics is circumscribed by available recording and intervention technologies. In particular, the ability to optically record and perturb dynamics in long-range, genetically-specified projections, and the ability to observe inter-regional and inter-laminar population dynamics at cellular resolution across large volumes of tissue, are necessary to understand how behaviorally-relevant dynamics emerge from cellular activity across the intact brain at multiple spatial scales in scattering tissue. Over the last 8 years new volumetric imaging methods have been developed to address this need.
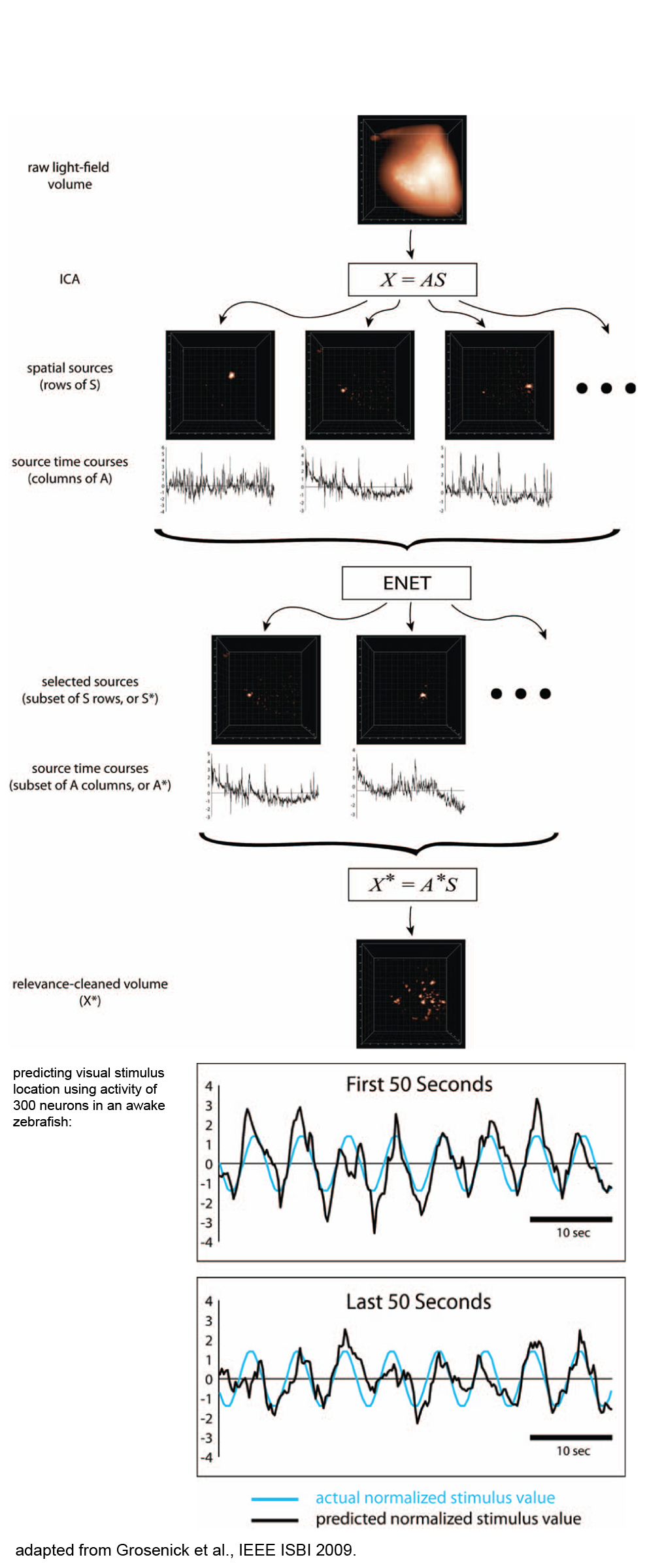 |
Light Field Microscopy for 3D functional neuroimagingThe invention of light field microscopy: Recording and control of the 4D light field in a microscope, with measurements of fluorescent bacteria in fixed mouse tissue, human hair highlights, and squid iridiphores: The first functional imaging at cellular resolution across a brain volume (in a transparent zebrafish during visual stimulation): Light field microscopy discussed as emerging functional imaging technique in Annual Reviews Neuroscience: Improving light field microscopy resolution using a wave-optics model for 3D deconvolution and application to a simple biological sample: Broxton M, Grosenick L, Yang S, Cohen N, Andalman A, Deisseroth K, and M Levoy. Optics Express (2013) 21(21) 25418–25439. First public demonstrations, discussion, and publication of light field microscopy and analytics during animal behavior, and demonstration of cellular-resolution functional imaging across brain regions in deep, scattering mammalian tissue:
Whole-animal imaging in C. elegans with light field microscopy (using the methods developed above in Levoy et al., 2006, Grosenick et al., 2009, and Broxton et al., 2013): Enhancing the performance of the light field microscope using wavefront coding. Cohen N, Yang SJ, Andalman AS, Broxton M, Grosenick L, Deisseroth, K and and M Levoy. Optics Express (2014) 22(20) 24817–24839. SPED Light Sheet Microscopy: Fast Mapping of Biological System Structure and Function. Tomer R, Lovett-Barron M, Kauvar I, Andalman A, Burns VM, Sankaran S, Grosenick L, Broxton M, Yang S, and K Deisseroth. Cell 163(7):1796–1806. Extended field-of-view and increased-signal 3D holographic illumination with time-division multiplexing. Yang SJ, Allen WE, Kauvar I, Andalman AS, Young NP, Kim CK, Marshel JH, Wetzstein G and K Deisseroth. Optics Express 23(25):32573–32581. Identification of cellular-activity dynamics across large tissue volumes in the mammalian brain Grosenick L*, Broxton M*, Kim CK*, Liston C*, Poole B, Yang SJ, Andalman A, Scharff E, Cohen N, Yizhar O, Ramakrishnan C, Ganguli S, Suppes P, Levoy M, and K Deisseroth. bioRxiv Preprint doi: https:doi.org10.1101132688. |
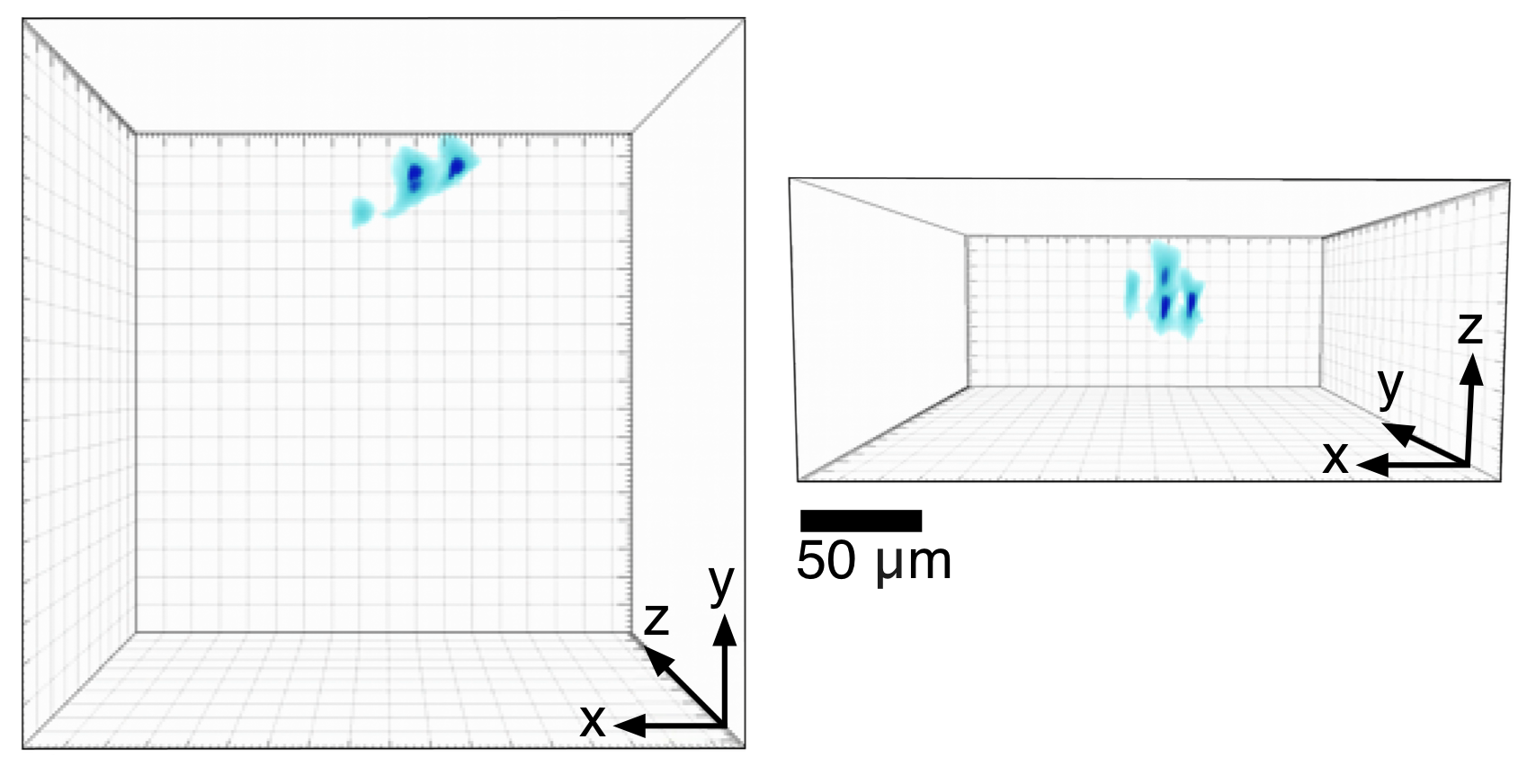 |
Light Field Illumination (LFI) for 3D optogenetic photostimulationUsing a digital micromirror device (DMD) and a microlens array, it is possible to generate structured volumes of light with kilohertz switching times. The basic optics and computation necessary for this technology, called Light Field Illumination (LFI) have been described previously. We are developing this technology for 3D photostimulation with optogenetics. This movie shows unpublished results (L.Grosenick, M. Broxton, K. Deisseroth) showing rapid formation of structured one-photon volumes for optical stimulation. Shown is a volume rendering of light field imaging reconstructions (Broxton, et al., 2013; reconstructed with 3x3x5 micron voxels) of a volume containing multiple localized peaks generated in a fluorescent volume (a hydrogel densely populated with sub-micron beads using light field illumination; imaging parameters: 40x 0.8; 20x 0.5NA objective; 125 micron pitch, 100% fill-factor rectangular microlens arrays; 1:0.7 demagnifying telecentric relay; DMD was a TI DLP9500 and illumination path used a matched tube lens and the same microlens array parameters; setup modeled on Levoy, et al., 2009). |